Decoding Product Security: Matthias Welzmiller, TRATON
On this episode of Decoding Product Security series, Gilad Bandel, VP Cybersecurity, hosted Matthias Welzmiller, Unit Head, Vehicle Cybersecurity and Safety, at TRATON (MAN). Gilad
" " indicates required fields

On this episode of Decoding Product Security series, Gilad Bandel, VP Cybersecurity, hosted Matthias Welzmiller, Unit Head, Vehicle Cybersecurity and Safety, at TRATON (MAN). Gilad
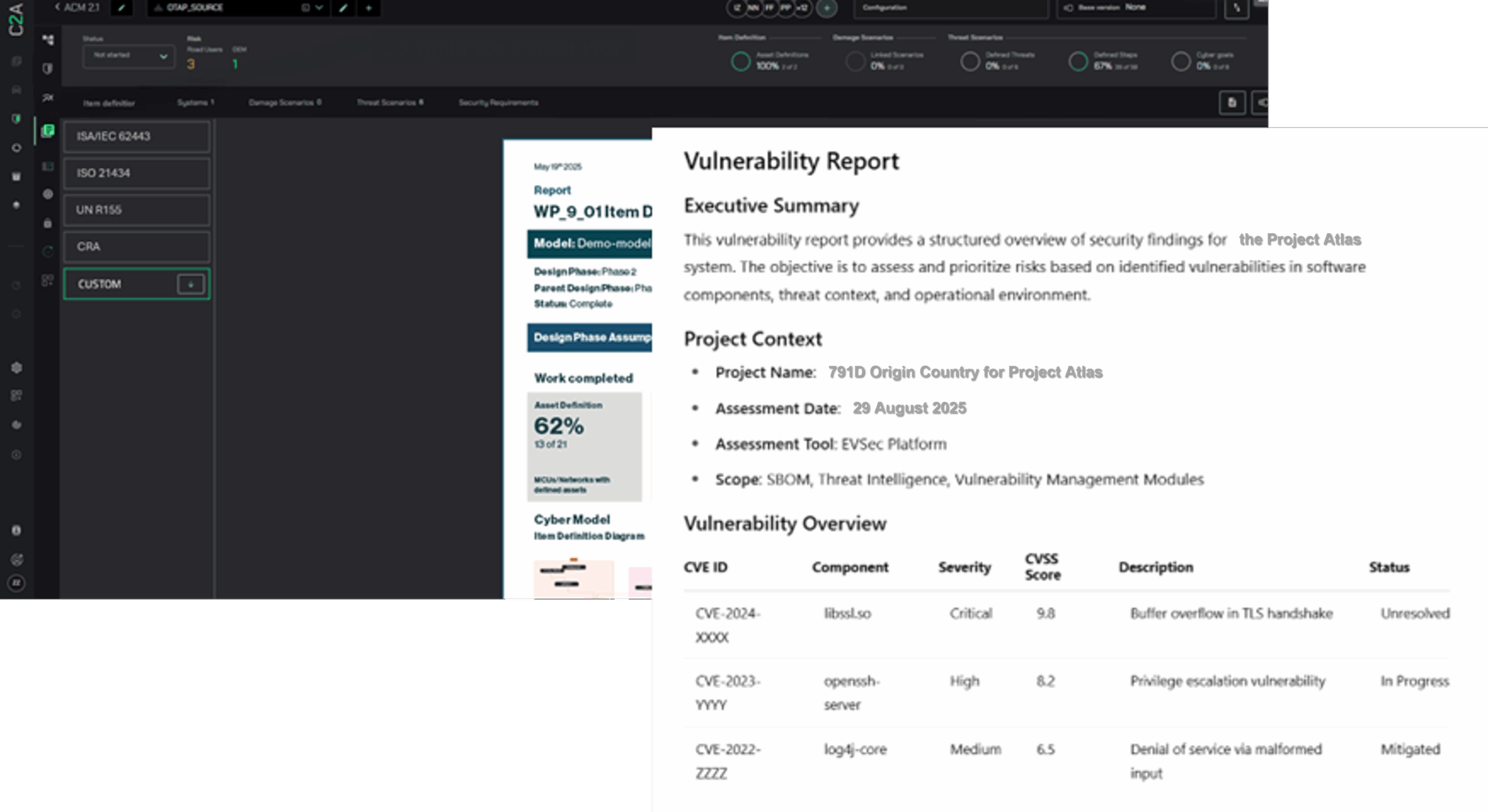
Rule 791D has officially arrived. Earlier this year, we covered the proposed ban on connected vehicle software and hardware manufactured in China and Russia. The

The new capability is designed to give organizations the tools and automation they need to meet regulatory requirements quickly, confidently, and without slowing innovation Jerusalem,

Why The New FDA Update Matters Right Now for MDMs The FDA’s Final Cybersecurity Guidance of June 2025 introduces significant new requirements under the FD&C

Introduction The EU’s Network and Information Systems Directive 2 (NIS2, Directive (EU) 2022/2555) came into effect in late 2024, establishing a new benchmark for cybersecurity

Dvir Reznik and Deloitte’s Partner Andreas Herzig discuss the impact of the Cyber Resilience Act on product security, supply chain challenges, and the evolving regulatory

On March 17, 2025, the new Department of Commerce (DoC) rule banning Chinese and Russian software from US vehicles, is entering into effect, and the

From Regulation to Implementation: How the CRA Forces the Industry to Reimagine Vulnerability Management (VM) OEMs in the automotive industry face an underlying threat in

The EU Cyber Resilience Act (CRA) officially became law on October 10, 2024, and was enacted on December 10, 2024. It sets a new cybersecurity

CRO
C2A Security

VP and GM, Medical Technology
C2A Security
Ken Zalevsky brings over 20 years of medical device cybersecurity experience to his role at C2A Security, where he serves as VP and GM, Medical Technology, following the acquisition of Vigilant Ops in October 2025. A former Bayer executive, Ken founded Vigilant Ops in 2019 after witnessing the consequences of inadequate technical preparation in the healthcare industry. He is an active contributor to CISA’s SBOM working groups and a frequent speaker on software supply chain security. Ken’s mission: transform SBOM from a compliance checkbox into operational intelligence that keeps patients safe while streamlining regulatory processes.